
Research
We offer purpose-built products, capabilities, and resources to simplify the research process, accelerate discovery, and address humanity’s most urgent needs.
Oracle products for researchers
Our products are designed to make research easier, faster, and more accurate. We want to empower researchers with the most-advanced technologies so they can unlock new insights and make discoveries that change the world.
AI for research
Oracle AI is a family of accelerated infrastructure, artificial intelligence, and machine learning (ML) services. For AI training and inferencing, Oracle’s AI infrastructure offers ultralow latencies for standalone graphics processing units (GPUs) and clusters with thousands of nodes. Using AI services, developers can add prebuilt models to applications and operations. With ML services, data scientists can build, train, and deploy models with their favorite open source frameworks or benefit from the speed of in-database machine learning.
Research AI services
- Anomaly detection service
- Data labeling
- Data science
- Digital assistant
- Forecasting
- Language
- Machine learning services
- Speech
- Virtual machines for data science
- Vision
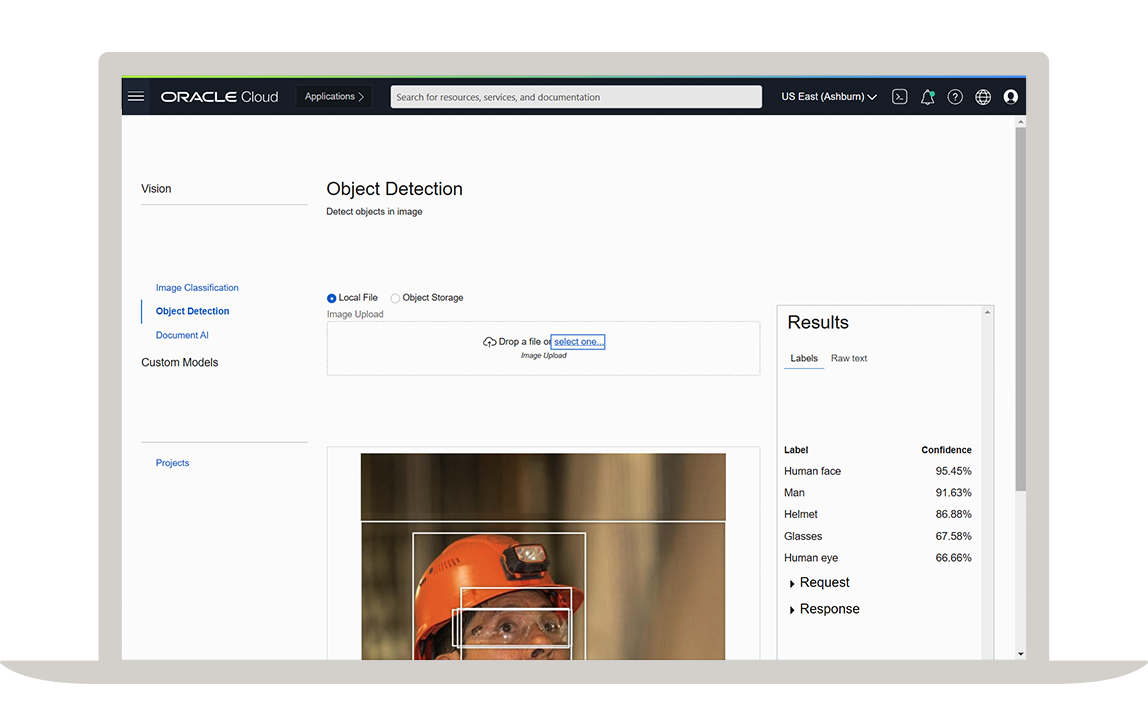
Process complex research workloads
Our purpose-built cloud for research scientists provides fast, flexible, and affordable compute capacity to fit any scientific workload, from flexible virtual machines (VMs) and high performance bare metal servers to lightweight containers and serverless computing.
Computing services for researchers
- Arm-based compute
- Bare metal servers
- Container Engine for Kubernetes
- Functions
- GPU-accelerated compute
- HPC compute
- Operating systems
- Virtual machines
- VMware
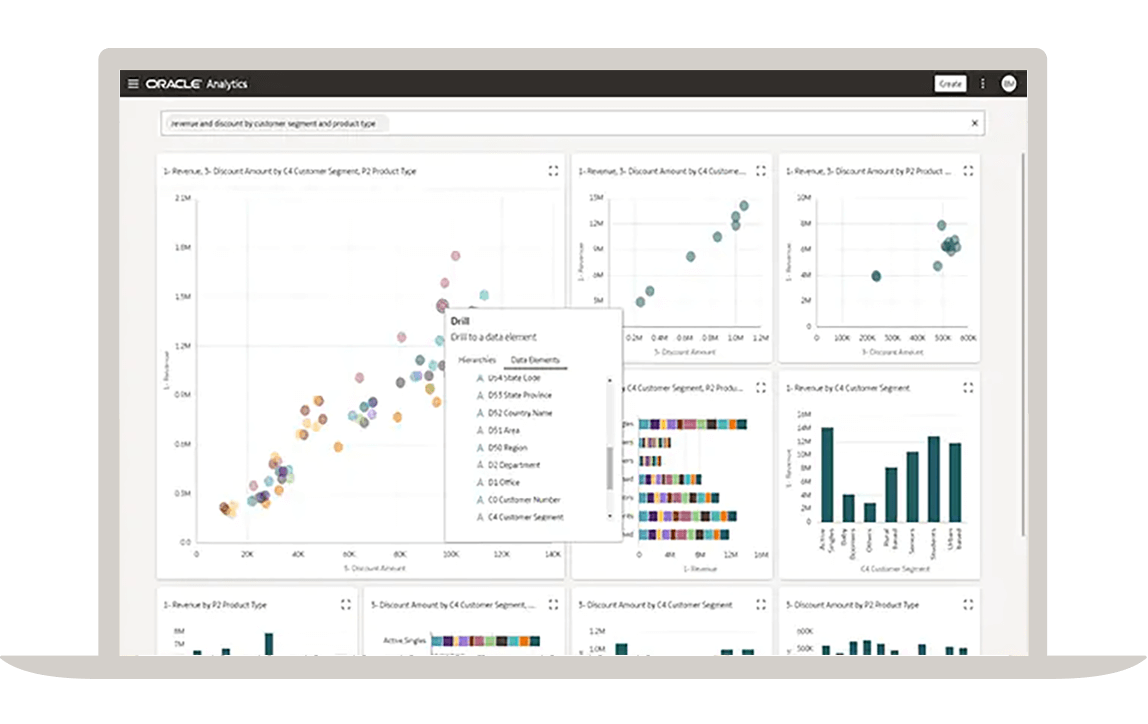
Summarize your collected data
Make sense of the data you collect with a service that provides the capabilities required throughout the entire research process. This includes data ingestion and modeling, data preparation, data visualization, and data collaboration. Quickly discover unseen patterns and insights from your studies.
Get more from your research with:
- Auto-insights
- Data quality insights
- Data modeling
- Data visualization
- Machine learning
- Mobile analytics
- Natural language processing
- Spatial analytics
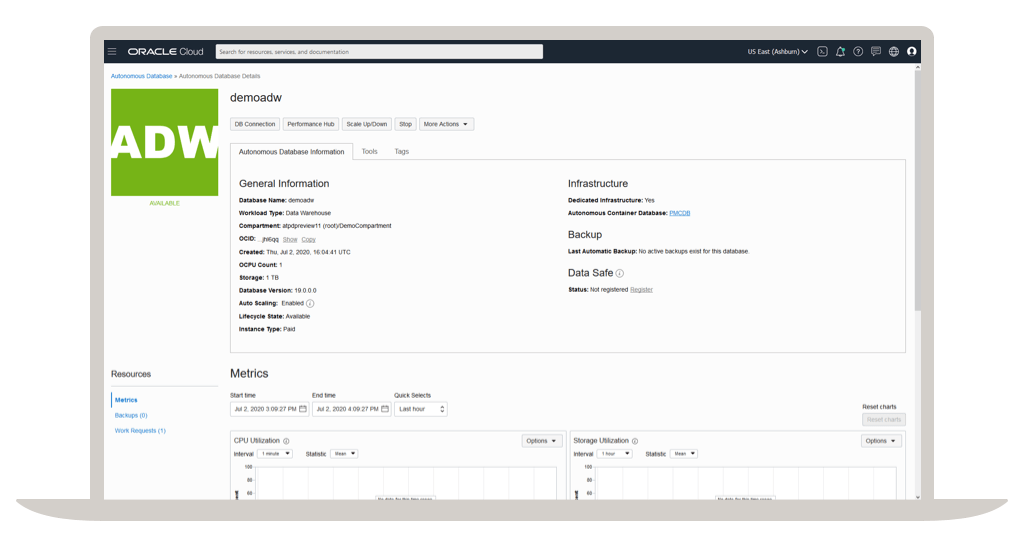
Store and retrieve your research
Oracle database services and products offer researchers cost-optimized and high performance versions of Oracle Database, the world's leading converged, multimodel database management system, as well as in-memory, NoSQL and MySQL databases. Oracle Autonomous Database, available on-premises via Oracle Cloud@Customer or on Oracle Cloud Infrastructure, enables researchers to simplify relational database environments and reduce management workloads.
Database offerings for researchers
- Autonomous Database
- Autonomous Data Warehouse
- Spatial database
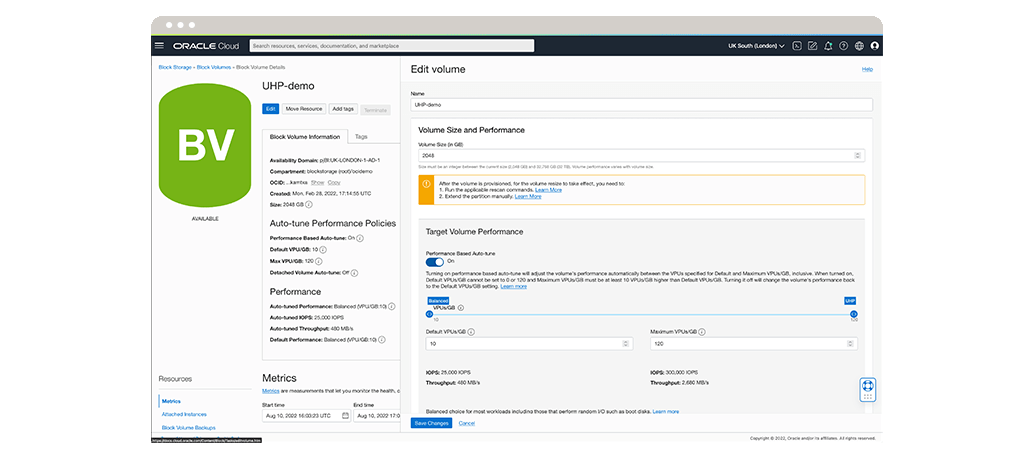
Securely store your research
Oracle Cloud Infrastructure provides extremely secure and low-cost cloud storage options. Through on-demand local, object, file, block, and archive storage, Oracle Cloud addresses key storage workload requirements and use cases. Researchers can use the storage gateway and data transfer service to safely and securely move their data to the cloud.
Cloud storage services
- Archive storage
- Boot/block volume
- File storage
- Local NVMe
- Object storage
- Storage gateway
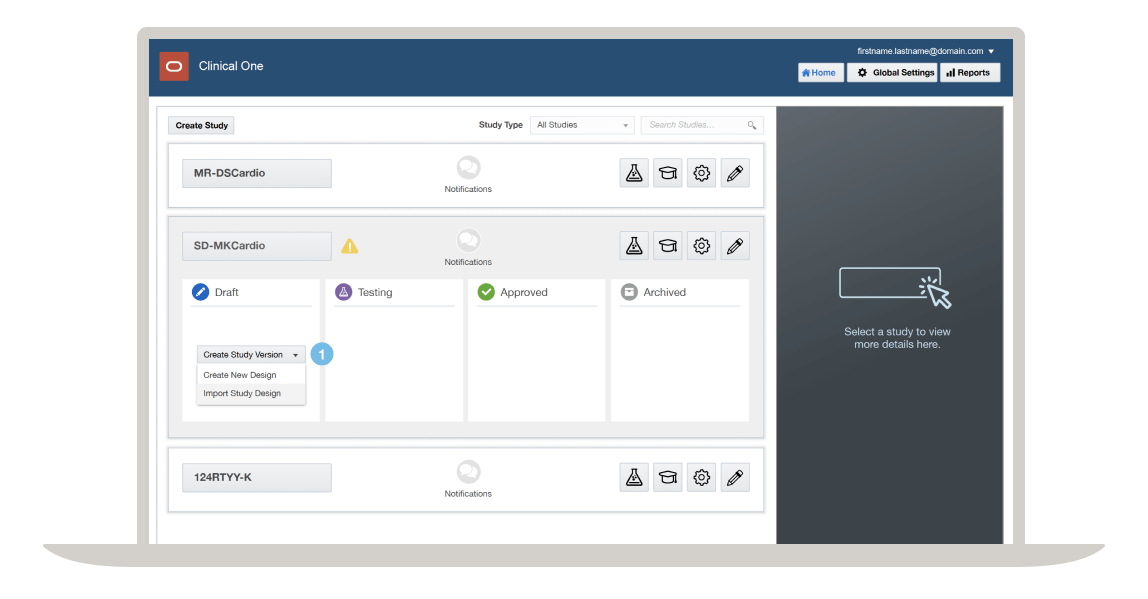
Clinical One unified platform
Clinical trials have been plagued with redundancy and inefficiency due to siloed data, fragmented technologies, and inconsistent operational practices. What if you could build a study once, enter data once, and do everything in a single place? Oracle Life Sciences Clinical One is the only platform that truly unifies people, processes, and data to simplify and accelerate clinical trials—today and in the future. Manage decision-making at scale in both clinical research and healthcare through the following:
Data management
Oracle Life Sciences Data Management Workbench is the only clinical trial data management solution that provides a validated, trustworthy single source of truth for all clinical trial data. The solution automatically reconciles data discrepancies and provides complete traceability. It also provides timely alerts about any issues in a trial, allowing you to take prompt action.
Trial management
Improve productivity by streamlining, automating, and reporting on clinical trial operations data across all clinical study management processes.
Data collection
Oracle Life Sciences Clinical One Data Collection allows you to collect datasets from various sources and consolidate them in a central location to gain valuable clinical research study insights.
Randomization and supplies management
Oracle Life Sciences Clinical One Randomization and Supplies Management can support randomization strategies for a wide range of research studies, from the simple to the complex, without requiring programming or coding. This eliminates the need for prolonged user acceptance testing.
Oracle real-world data solutions, fueled by Oracle Learning Health Network
We have a nationwide network of diverse health systems that fuels and shares de-identified data to help accelerate the discovery, development, and deployment of groundbreaking insights and therapies. Our more than 100 members conduct high-volume clinical trials and outcomes research. We provide one of the largest real-world data sets and help members launch clinical trials. For health systems that don’t currently have clinical trial capabilities, we can supply the necessary technology and training through our collaboration with Elligo.
Our members can:
- Activate, execute, and accelerate their clinical studies
- Access our national de-identified real-world data set to conduct observational studies and post-market surveillance
- Access peer research and share learnings with a network of participants
- Get help on activating clinical trials, enrolling patients, and removing obstacles
- Gain opportunities to launch or contribute to industry trials and keep more patients in network
Trusted safety case management
Oracle Life Sciences Argus is the market-leading, trusted solution for processing, analyzing, and reporting adverse event cases originating in premarket and post-market drugs, biologics, vaccines, devices, and combination products. Transform your case management efficiency with innovative capabilities, such as built-in automation, workflow optimization, and conditional touchless processing.
Workflow automation
The built-in automation features in Argus can significantly reduce manual work by 50% or more. These features include intake, case prioritization, field validations, letters, action items, coding, listedness, narratives, case lock, submission, and archiving.
Regulatory compliance
Argus is a dependable solution that complies with drug, vaccine, and device regulations and standards worldwide, including E2B(R3), E2B(R2), eVAERS, eMDR, MIR, and IDMP.
Safety analytics
Oracle Life Sciences Argus Advanced includes Oracle Analytics, which is a powerful and flexible AI-powered solution that enables users to discover new insights and make informed business decisions more quickly.
Leading-edge pharmacovigilance science
Oracle Life Sciences Empirica is a solution that’s widely recognized for its ability to detect, analyze, and manage safety signals in various products, including drugs, biologics, vaccines, devices, and combination products in both pre- and post-market settings. Oracle is constantly pushing the boundaries of innovation, tirelessly exploring and perfecting state-of-the-art data mining algorithms and statistical techniques to power Empirica.
Safety signal management
- See what health authorities such as the FDA see.
- Comply with EU GVP Module IX and CIOMS VII.
- Avoid investing in unsafe products with early risk detection.
- Use a powerful data mining engine with best-in-class algorithms.
- Gain flexible signal management analytics with rich visualizations.
- Reduce statistical noise for fewer false positives and less work.
Smarter signal management
Safety teams responsible for signal management are encountering new challenges, including identifying hidden signals, evaluating data from multiple large datasets, and improving safety information prior to the start of clinical research and trials. Please refer to our ebook for information on the latest advancements in methodologies and technologies, including artificial intelligence, that can help address these challenges.
Oracle Health Sciences ClearTrial Cloud Service product features
Portfolio planning
Leverage embedded industry intelligence, clinical knowledge, and activity-based planning to optimize your clinical research study planning and sourcing.
Clinical trial planning
The end-to-end planning process can be reduced from weeks to hours with visibility into industry standards, detailed tasks, timelines, costs, and resources.
Accurate study budgets
Get insight into the operational and financial planning for a study to negotiate fair market value effectively using industry benchmarks.
Shorten RFP and bid management cycles
Generate clinical study request for proposal (RFP) specifications based on a minimal number of clinical assumptions and reduce the disruptive nature and impact of change orders.
Optimize clinical trial and portfolio management
To maximize efficiency and minimize expenses, it’s important to implement a centralized and standardized approach for trials across the portfolio.
Solution brief: ClearTrial - Advanced visibility for portfolio planning (PDF)
Better-quality safety case processing
Oracle Life Sciences Safety One Intake uses artificial intelligence to improve the speed, quality, and compliance of safety case processing and help users manage the ever-growing volume of adverse events. Safety One Intake offers the following capabilities and benefits:
- Automatically extracts adverse event data from any source documents
- Uses artificial intelligence, machine learning, and natural language processing
- Leverages Oracle’s considerable expertise and experience in AI and safety
- Significantly reduces manual data entry, allowing staff to focus on high-value activities
- Delivers accuracy and speed, proven in customer pilot projects
- Facilitates the review of results with an intuitive user interface
Oracle Life Sciences Site Select
Oracle Life Sciences Site Select
Site Select offers a data-driven method for assessing selection and performance factors to help identify the sites and target populations that are best suited for studies so you can open sites on time and meet enrollment targets.
Oracle Life Sciences Site Activate
Site Activate enables sponsors, CROs, and sites to get a clinical study started in the shortest time possible. Site Activate workflows help study teams complete and track the specific documents and tasks necessary for any site, country, or study based on the relevant regulatory and SOP requirements.
Oracle Life Sciences Site Analyze Cloud Service
Site Analyze presents timely status updates across your studies and insights to streamline operational processes. Our visually rich analytics and dashboards help your team transition from a reactive process to a proactive one.
Oracle Life Sciences Analyze Insight for Activate
Analyze Insight for Activate improves transparency, communication, and collaboration with CROs by providing sponsors with timely status updates and operational insights into outsourced studies with their CRO partners, leading to enhanced performance and governance. Through visually rich analytics and dashboards, we help transition your team from a reactive process to a more proactive one.
Read the Accelerating Clinical Trials brochure (PDF)
Customer success stories
Our world-class researchers are helping make the world a better place. Hear more from our research customers to learn why Oracle was the right choice for their projects, and why we'll be the right partner for yours too.

“Without the Oracle compute resources, the research that we're carrying out could have taken months to years to complete. With Oracle behind us, we got access to a large number of GPU compute nodes, so we could complete the research in a matter of only weeks”
Oracle products for research institutes








